A second order reaction can be altered to a first order reaction by taking one of the reactant in large excess, such reaction is called pseudo first order reaction. Let us consider the acid hydrolysis of an ester,
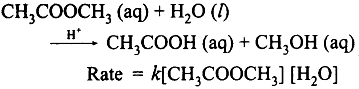
If the reaction is carried out with the large excess of water, there is no significant change in the concentration of water during hydrolysis, i.e., the concentration of water remains almost constant.
Now, we can define k [H2O] = k’ ; Therefore the above rate equation becomes
Rate = k’ [CH3COOCH3]
Thus it follows first-order kinetics.